Scrosati: The clonal seaweed Chondrus crispus as a foundation species
ABSTRACT
The clonal seaweed Chondrus crispus (Rhodophyta, Gigartinales) forms extensive stands at low intertidal elevations on wave-sheltered rocky shores of the North Atlantic. This study investigates if this bushy alga acts as a foundation species in such habitats. The abundance (percent cover) of C. crispus, all other algae, and invertebrates was measured in 390 quadrats spanning 350 km of coast in Nova Scotia, Canada. In these low-intertidal habitats, fucoid algae are the largest organisms and can form extensive canopies, but their cover was unrelated to benthic species richness and to C. crispus cover. Species richness, however, increased with C. crispus cover from low to intermediate cover values, showing little change towards full C. crispus cover. Species composition (a combined measure of species identity and their relative abundance) differed between quadrats with low (0–1%) and high (60–100%) cover of C. crispus. High C. crispus cover was associated to more invertebrate species but fewer algal species than low C. crispus cover. However, the average abundance of algal and invertebrate species occurring in both cover groups was often higher under high C. crispus cover, contributing to a higher average richness at the quadrat scale. Overall, only 16% of the observed variation in species richness was explained by C. crispus cover. Therefore, this study suggests that C. crispus acts as a foundation species but with a moderate influence.
Key words: Chondrus; community structure; foundation species; Gigartinales; intertidal
INTRODUCTION
Foundation species are organisms that create habitats by providing substrate through their body structures and that modify local environmental conditions ( Altieri and van de Koppel 2014). Through those mechanisms, foundation species influence the richness (number of species) and composition (a combined measure of the identity and relative abundance of species) of local communities. Terrestrial examples of foundation species are alpine plants, which protect smaller plants from wind and cold stress ( Ballantyne and Pickering 2015), bromeliads, which host microfauna in rainwater-filled phytotelmata ( Hammill et al. 2015), and trees, which alter microclimates and host fungi, lichens, plants, and animals on trunks and branches ( Thomas and Packham 2007, Bäcklund et al. 2016, Dislich and Mantovani 2016). In marine intertidal habitats, mussels and barnacles can limit local abiotic stress through their shells and serve as complex substrate for colonization, increasing local richness ( Harley and O’Riley 2011, Arribas et al. 2014). Intertidal seaweed canopies can increase local richness by limiting understory abiotic stress during low tides ( Watt and Scrosati 2013a) and by providing habitat for epiphytic algae and invertebrates ( Johnson and Scheibling 1987, Longtin et al. 2009, Jones and Thornber 2010). The seaweed Chondrus crispus Stackhouse (Rhodophyta, Gigartinales) is a common species at low intertidal elevations on cold-temperate North Atlantic shores ( Lubchenco and Menge 1978, Scrosati and Heaven 2007, Hu et al. 2010). Because of its ecological and economic importance, it is a well studied species. Thus, knowledge exists on its life cycle ( Chen and McLachlan 1972), reproductive phases ( Scrosati et al. 1994, Scrosati and Mudge 2004, Guidone and Grace 2010, Garbary et al. 2011), biomechanics ( Carrington et al. 2001), interspecific interactions ( Lubchenco and Menge 1978, Lubchenco 1980), cultivation ( Chopin et al. 1999, Matos et al. 2006), and bioremediation potential ( Corey et al. 2013). It is a clonal seaweed that can form extensive intertidal beds, as the holdfasts can grow across the substrate producing fronds (ramets) in high densities ( McLachlan 1991). These dense stands can be considered as turfs ( Connell et al. 2014). Therefore, this seaweed has the potential to act as a foundation species. However, little is known in this regard. For this reason, this paper examines how the abundance (cover) of intertidal C. crispus relates to the structure of associated communities. The first hypothesis was that the species richness of the associated communities would increase with C. crispus cover, as habitat complexity increases in that way ( Matias et al. 2015). On the other hand, as species composition is more sensitive than richness to changes in habitat conditions ( Heaven and Scrosati 2008), this study also tested the hypothesis that the composition of associated communities would change with C. crispus cover.
MATERIALS AND METHODS
This paper uses an existing dataset that describes the percent cover of intertidal species measured along 350 km of coastline between Taylor Head (44°48.79′ N, 62°34.26′ W) and Godie Point (45°36.69′ N, 60°49.19′ W), on the Atlantic coast of Nova Scotia, Canada, in May 2008. The full dataset has been published by Watt and Scrosati (2014). The data were originally gathered to investigate the effects of the cover of fucoid algal canopies ( Ascophyllum nodosum (Linnaeus) Le Jolis and Fucus spp.) on the species richness and composition of benthic communities at the high (1–1.5 m of elevation above chart datum), middle (0.5–1 m), and low (0–0.5 m) intertidal zones of wave-sheltered rocky shores ( Watt and Scrosati 2013b). C. crispus was absent at the high zone and rare at the middle zone (mean percent cover = 1%). However, at the low zone, C. crispus had a mean percent cover of ~20%, with a range of 0–100% ( Watt and Scrosati 2014), forming canopies that were only smaller than those of the fucoid algae, which covered all other species when abundant. At the low intertidal zone, fucoid algal cover did not affect species richness or composition, likely because fucoid algae have little environmental influences during the short low tides that characterize low elevations ( Watt and Scrosati 2013a, 2013b). Using the data published by Watt and Scrosati (2014), it can also be shown that fucoid algal cover was also unrelated to the cover of C. crispus at the low intertidal zone (r = 0.009, p = 0.862, n = 390 quadrats) ( Fig. 1). However, C. crispus might still influence local communities at the low intertidal zone, because its stands, despite being shorter than those of fucoid algae, are more compact and thus increase habitat complexity ( Fig. 2). Therefore, the two hypotheses of this study were tested using the data published by Watt and Scrosati (2014) only for the low intertidal zone. Specifically, the data used for this paper describe the percent cover of all invertebrates and algae (including C. crispus but excluding the fucoid algae) measured during low tides in 390 random quadrats (20 cm × 20 cm) using a metallic frame divided in 100 squares with monofilament line. For each quadrat, the percent cover of each species was determined by counting the number of squares in which the species’ cover was 50% or more. If a species was present in a quadrat, but covered less than 1% in total, its cover was recorded as 0.5%. Because fucoid canopies, whenever present, covered all other organisms, the percent cover of C. crispus and the associated species was measured after carefully moving away the fucoid canopies. Organisms were identified with field guides ( Gibson 2003, Martinez 2003, Sept 2008) and taxonomic keys ( Pollock 1998, Sears 1998) to as low a taxonomic level as possible, as normally done in studies that evaluate primary producers and consumers ( Valdivia and Molis 2009, Golléty et al. 2011). As most of the encountered organisms were identified to the species level ( Table 1), the text refers to species richness and composition for ease of presentation. For each quadrat, the richness of associated species was calculated as the number of species (excluding C. crispus) found in the quadrat. The relationship between C. crispus cover and species richness was evaluated through linear and quadratic regression analyses ( Sokal and Rohlf 2012). As the linear and quadratic models differ in number of parameters, the models were compared using their respective values of the corrected Akaike’s information criterion (AICc). Using both AICc values, the weight of evidence was calculated for each model. The plausibility of the quadratic model relative to the linear one was evaluated by calculating the evidence ratio, that is, the ratio between the weight of evidence for the quadratic model and the weight of evidence for the linear model ( Anderson 2008). Once the best of the two models (the one with the lowest AICc value) was identified, its adjusted squared correlation coefficient (R 2) was calculated to determine the amount of variation in richness that could be explained by C. crispus cover ( Sokal and Rohlf 2012). These analyses were done with PRISM 6.0c for Mac OS (GraphPad, La Jolla, CA, USA). Since species richness differed between stands with low and high cover of C. crispus (see results), species composition was compared between two groups that represented that difference well. The low-cover group included the quadrats that had 0–1% cover of C. crispus, intended to represent null to small C. crispus influences. The high-cover group included the quadrats that had 60–100% cover of C. crispus, intended to represent high C. crispus influences considering a cover range along which richness varied little (see results). Species composition was compared between these two groups through a multivariate analysis of similarity (ANOSIM) based on Bray-Curtis distances among quadrats using fourth-root-transformed data to limit the influence of abundant species in the analysis ( Clarke and Warwick 2001). The differences between both groups were visually summarized using non-metric multidimensional scaling (NMDS). These analyses were done with PRIMER 6.1.11 for Windows (PRIMER-E, Lutton, UK).
RESULTS
A total of 69 species (25 algae excluding C. crispus and fucoid algae, one lichen, and 43 invertebrates) were identified on the studied coast ( Table 1). The quadratic model relating the percent cover of C. crispus and the richness of associated species (AICc = 1,953.7) was better than the linear model (AICc = 1,969.3). The ratio between the values of weight of evidence for the quadratic (0.9995904) and linear (0.0004096) models indicated that the quadratic model was ~2,441 times more plausible than the linear model. Therefore, the quadratic function was used to examine the relationship ( Fig. 3) between C. crispus cover ( C) and species richness ( S): This function predicts that species richness increases from a minimum value of ca. 11 species under 0% cover towards higher cover values, exhibiting signs of saturation at intermediate cover values and reaching a peak of ca. 15 species under 66% cover, after which it decreases slightly towards 100% cover ( Fig. 3). The adjusted R 2 value for the quadratic model was 0.156 (p < 0.001), indicating that 15.6% of the observed variation in species richness could be explained by changes in C. crispus cover. In the group of quadrats with 0–1% cover of C. crispus, 57 species (22 algae, one lichen, and 34 invertebrates) were identified, while 54 species (13 algae, one lichen, and 40 invertebrates) were identified in the group of quadrats with 60–100% cover ( Table 1). Both cover groups shared 42 species (61%) of the total number encountered in this study ( Table 1). At the quadrat scale, species composition differed significantly between low (0–1%) and high (60–100%) cover of C. crispus (ANOSIM, global R = 0.461, p < 0.001). The difference in composition between both cover groups was clearly revealed by NMDS ordination ( Fig. 4).
DISCUSSION
This study suggests that the clonal seaweed C. crispus acts as a foundation species at low elevations in wave-sheltered rocky intertidal habitats in Atlantic Canada. Its influence appears to be mild, however, because only 15.6% of the observed variation in the richness of associated species could be predicted from C. crispus cover. Moreover, the positive influence of cover was most evident only from low to intermediate cover values. From intermediate to high cover values, species richness changed little, showing a slight decrease towards full cover. Such a limited change in richness suggests that intermediate values of C. crispus cover allows for the occurrence of all species from the species pool available at low elevations. A similar pattern was found for fucoid algal canopies at middle and high elevations, which is where fucoid canopies influence local species richness ( Watt and Scrosati 2013a). The community influence of C. crispus likely relates to its effects on abiotic stress and habitat complexity. During low tides, exposure to the air causes desiccation and thermal stress in intertidal organisms ( Raffaelli and Hawkins 1999), but dense algal stands (like those of C. crispus) retain moisture and shade habitats at low tide, limiting such stresses ( Hay 1981, Scrosati and DeWreede 1998, Guenther and Martone 2014, Smith et al. 2014, Fernández et al. 2015). Except for a few sessile invertebrates (barnacles, Semibalanus balanoides, and mussels, Mytilus spp.), intertidal invertebrates are often mobile and have little tolerance for low-tide stresses, suggesting that their occurrence could be enhanced in C. crispus stands. During high tides, dense stands of C. crispus limit water velocity within the stands, limiting hydrodynamic stress and providing a safer environment for mobile invertebrates than bare rock ( Johnson 2001, Boller and Carrington 2006). Finally, C. crispus increases habitat complexity because its fronds are branched and occur in dense stands ( McLachlan 1991) ( Fig. 2). Such structures create environmental mosaics in small volumes, potentially allowing for small species with different niche requirements to coexist ( Matias et al. 2015). In support of the above notions, the group of high-cover quadrats had six more invertebrate species than the group of low-cover quadrats. Moreover, 65% of the invertebrate species that occurred in both cover groups were more abundant under high cover ( Table 1). Increases in invertebrate abundance with the stand biomass of morphologically complex intertidal seaweeds have recently been described for polar shores ( Martín et al. 2016). The possible mechanisms discussed above might have less influence on the associated algae, as algae compete for substrate, light, and nutrients with C. crispus. In fact, the group of low-cover quadrats had, in total, nine more algal species than the group of high-cover quadrats ( Table 1). However, at the quadrat scale, such a difference did not prevent high-cover quadrats from having a higher average richness of associated species than low-cover quadrats. Moreover, of the algal species that occurred in both cover groups, 60% were more abundant under high C. crispus cover ( Table 1). Therefore, it seems that, while a high cover of C. crispus may prevent some algae from being present, many algal and invertebrate species are actually favoured by high cover, contributing to a higher average richness at the quadrat scale. In addition, high C. crispus cover favoured a higher compositional similarity among quadrats than low cover, as indicated by the lower spatial dispersion of high-cover quadrats in the NMDS ordination, compared with low-cover quadrats. This difference also occurs in alpine plant communities that are influenced by cushion plants, as these foundation species increase the compositional similarity among stands ( Kikvidze et al. 2015). The effects of fucoid algal canopies on individual species have been studied for some cases ( Bertness et al. 1999). However, the effects of C. crispus, especially on the many small species that occur in its intertidal stands, remain largely unstudied. Therefore, the mechanisms that possibly explain the results of this study (summarized above) should be evaluated experimentally. Another aspect that will require experimental study is the understanding of the observed variation in species richness. The correlation analysis showed that 84.4% of the variation in richness observed at the low intertidal zone was influenced by factors other than C. crispus cover. Such factors could include substrate slope ( Harley 2008, Vaselli et al. 2008) and rugosity ( Menge et al. 2010), local differences in water flow ( Lau and Martinez 2003), and interspecific interactions ( Menge 1995, Kim 1997, Ellrich et al. 2015), which could be evaluated with field experiments. In some communities, large foundation species (e.g., trees) facilitate the occurrence of smaller foundation species (e.g., bromeliads) that in turn favour the occurrence of other species ( Angelini and Silliman 2014). Such a facilitation cascade does not seem to occur between the large canopy-forming fucoid algae ( A. nodosum and Fucus spp.) and C. crispus on wave-sheltered shores in Atlantic Canada. These fucoid algae increase local species richness only at high and middle intertidal elevations, where their canopies greatly limit thermal and desiccation extremes during low tides ( Beermann et al. 2013, Watt and Scrosati 2013a). However, C. crispus is almost absent at such elevations ( Watt and Scrosati 2014). It is at low elevations where local species richness increases with C. crispus cover, but the fucoid algae appear not to affect C. crispus cover there ( Fig. 1). Therefore, while the fucoid algae are foundation species at high and middle elevations, C. crispus seems to be a foundation species, albeit less markedly, at low elevations. The field manipulation of C. crispus cover to evaluate effects on the richness and composition of associated communities could strengthen this study’s conclusion. Given the high quadrat replication and large geographic extent of this mensurative study, a field experiment would likely yield the same basic results. For example, the large-scale mensurative study that evaluated fucoid algal effects on intertidal communities ( Watt and Scrosati 2013b) was supported by a manipulative field experiment that asked the same question but could only be done at one location due to logistics ( Watt and Scrosati 2013a). Large-scale mensurative studies like this one are valuable because they provide conclusions with a high degree of realism that are representative of a region of interest ( Sagarin and Pauchard 2010).
ACKNOWLEDGEMENTS
I appreciate the constructive comments received from two anonymous reviewers. This study was funded by a Discovery Grant awarded to the author by the Natural Sciences and Engineering Research Council of Canada (NSERC).
Fig. 1
Lack of relationship between the percent cover of canopy-forming fucoid algae (Ascophyllym nodosum and Fucus spp.) and the percent cover of Chondrus crispus. 
Fig. 2
Low intertidal zone viewed at low tide, showing Chondrus crispus (C) and fucoid algae (F). Scale bar represents: 15 cm. 
Fig. 3
Quadratic relationship between the percent cover of Chondrus crispus and the species richness of the associated communities. 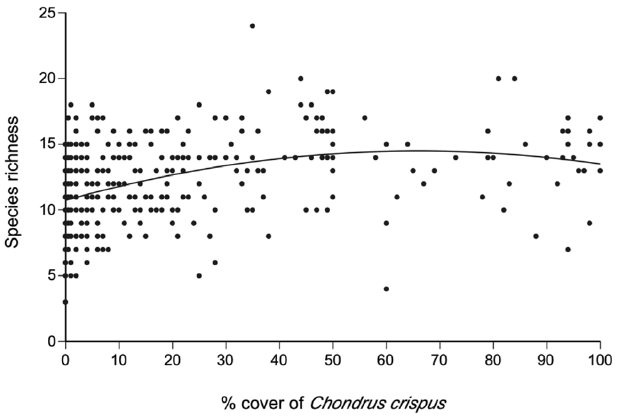
Fig. 4
Non-metric multidimensional scaling summarizing the difference in species composition between quadrats with low (0–1%) and high (60–100%) percent cover of Chondrus crispus. 
Table 1
Abundance of species at the low intertidal zone of wave-sheltered rocky shores in Nova Scotia under low (0–1%) and high (60–100%) percent cover of Chondrus crispus
|
Low cover of C. crispus (n = 110) |
High cover of C. crispus (n = 37) |
|
Algae |
|
Bangia atropurpurea (Mertens ex Roth) C. Agardh |
0.31 ± 0.21 |
0 |
|
Calothrix spp. |
< 0.01 |
0 |
|
Ceramium spp. |
0.03 ± 0.01 |
0.15 ± 0.16 |
|
Chaetomorpha spp. |
0 |
0.04 ± 0.02 |
|
Chordaria flagelliformis (O. F. Müller) C. Agardh |
0.31 ± 0.23 |
0 |
|
Cladophora rupestris (Linnaeus) Kützing |
0.18 ± 0.16 |
0.32 ± 0.17 |
|
Cladophora spp. |
0.13 ± 0.09 |
0.07 ± 0.03 |
|
Corallina officinalis Linnaeus |
0.58 ± 0.14 |
8.45 ± 1.73 |
|
Cystoclonium spp. |
0.01 ± 0.01 |
0.09 ± 0.04 |
|
Devaleraea ramentacea (Linnaeus) Guiry |
0.01 ± 0.01 |
0.03 ± 0.03 |
|
Dumontia contorta (S. G. Gmelin) Ruprecht |
0.59 ± 0.29 |
0.32 ± 0.14 |
|
Elachista chondrii Areschoug |
0 |
0.01 ± 0.01 |
|
Elachista spp. |
0.05 ± 0.05 |
0 |
|
Hildenbrandia rubra (Sommerfelt) Meneghini |
3.50 ± 0.69 |
0.54 ± 0.10 |
|
Laminaria digitata (Hudson) J. V. Lamouroux |
0.01 ± 0.01 |
0 |
|
Lithothamnion glaciale Kjellman |
1.75 ± 0.44 |
0.71 ± 0.24 |
|
Palmaria palmata (Linnaeus) Weber & Mohr |
0.15 ± 0.15 |
0 |
|
Petalonia fascia (O. F. Müller) Kuntze |
< 0.01 |
0 |
|
Polysiphonia spp. |
0.02 ± 0.02 |
0 |
|
Ralfsia spp. |
0.09 ± 0.06 |
0 |
|
Rhodochorton purpureum (Lightfoot) Rosenvinge |
0.10 ± 0.07 |
0 |
|
Rhodomela confervoides (Hudson) P. C. Silva |
0.06 ± 0.05 |
0.14 ± 0.14 |
|
Scytosiphon spp. |
0.05 ± 0.02 |
0 |
|
Spongomorpha aeruginosa (Linnaeus) Hoek |
0 |
0.03 ± 0.02 |
|
Ulva lactuca Linnaeus |
< 0.01 |
0 |
|
Lichen |
|
Verrucaria maura Wahlenberg |
3.41 ± 0.67 |
0.59 ± 0.31 |
|
Invertebrates |
|
Alcyonidium spp. |
0 |
0.01 ± 0.01 |
|
Anomia simplex (d’Orbigny 1853) |
0.04 ± 0.01 |
0.31 ± 0.05 |
|
Asterias rubens (Linnaeus 1758) |
0.02 ± 0.01 |
0.28 ± 0.13 |
|
Astyris lunata (Say 1826) |
0 |
0.04 ± 0.02 |
|
Bryozoa |
0.07 ± 0.03 |
0.88 ± 0.33 |
|
Cancer irroratus (Say 1817) |
0.04 ± 0.02 |
0 |
|
Carcinus maenas (Linnaeus 1758) |
0.02 ± 0.01 |
0.09 ± 0.06 |
|
Clava multicornis (Forsskål 1775) |
0.08 ± 0.02 |
0 |
|
Crenella glandula (Totten 1834) |
0.02 ± 0.01 |
0.04 ± 0.02 |
|
Dynamena pumila (Linnaeus 1758) |
1.17 ± 0.38 |
0.64 ± 0.12 |
|
Electra pilosa (Linnaeus 1767) |
0 |
0.35 ± 0.11 |
|
Flustrellidra hispida (O. Fabricius 1780) |
0.06 ± 0.02 |
0.38 ± 0.08 |
|
Gammarus spp. with biramous uropod |
0.23 ± 0.02 |
0.24 ± 0.04 |
|
Gammarus spp. with uniramous uropod |
0.16 ± 0.02 |
0.13 ± 0.04 |
|
Halacaridae |
0.01 ± 0.01 |
0.01 ± 0.01 |
|
Halichondria (Halichondria) bowerbanki (Burton 1930) |
0 |
0.16 ± 0.07 |
|
Halichondria (Halichondria) panicea (Pallas 1766) |
< 0.01 |
0.11 ± 0.06 |
|
Halisarca spp. |
0.07 ± 0.07 |
0.27 ± 0.17 |
|
Harmothoe imbricata (Linnaeus 1767) |
0 |
0.01 ± 0.01 |
|
Hiatella arctica (Linnaeus 1767) |
0 |
0.03 ± 0.02 |
|
Idotea balthica (Pallas 1772) |
0.03 ± 0.01 |
0.05 ± 0.02 |
|
Idotea phosphorea (Harger 1873) |
< 0.01 |
0.01 ± 0.01 |
|
Jaera marina (Fabricius 1780) |
0.08 ± 0.02 |
0.16 ± 0.04 |
|
Lacuna vincta (Montagu 1803) |
0.26 ± 0.02 |
0.44 ± 0.03 |
|
Leucosolenia botryoides (Ellis & Solander 1786) |
0.01 ± 0.01 |
0.07 ± 0.03 |
|
Littorina littorea (Linnaeus 1758) |
1.14 ± 0.15 |
1.09 ± 0.14 |
|
Littorina obtusata (Linnaeus 1758) |
0.27 ± 0.02 |
0.14 ± 0.04 |
|
Margarites costalis (Gould 1841) |
0.06 ± 0.01 |
0.27 ± 0.04 |
|
Membranipora spp. |
0.04 ± 0.01 |
0.42 ± 0.06 |
|
Mytilus spp. |
0.24 ± 0.02 |
0.15 ± 0.04 |
|
Nematoda |
0.09 ± 0.02 |
0.18 ± 0.04 |
|
Nemertea |
0 |
0.03 ± 0.02 |
|
Nucella lapillus (Linnaeus 1758) |
0.39 ± 0.10 |
0.05 ± 0.03 |
|
Obelia spp. |
0.30 ± 0.06 |
0.10 ± 0.04 |
|
Oligochaeta |
0.03 ± 0.01 |
0.04 ± 0.02 |
|
Ophiopholis aculeate (Linnaeus 1767) |
0 |
0.01 ± 0.01 |
|
Semibalanus balanoides (Linnaeus 1767) |
2.60 ± 0.58 |
0.22 ± 0.07 |
|
Spirorbis borealis (Daudin 1800) |
< 0.01 |
0.04 ± 0.02 |
|
Spirorbis spirorbis (Linnaeus 1758) |
0.22 ± 0.02 |
0.24 ± 0.04 |
|
Strongylocentrotus droebachiensis (O. F. Müller 1776) |
0.02 ± 0.02 |
0 |
|
Testudinalia testudinalis (O. F. Müller 1776) |
0.29 ± 0.03 |
0.26 ± 0.04 |
|
Tonicella rubra (Linnaeus 1767) |
0 |
0.01 ± 0.01 |
|
Urosalpinx cinerea (Say 1822) |
0.02 ± 0.01 |
0.01 ± 0.01 |
REFERENCES
Altieri, AH. & van de Koppel, J. 2014. Foundation species in marine ecosystems. In : Bertness MD, Bruno JF, Silliman BR, Stachowicz JJ, editors Marine Community Ecology and Conservation. Sinauer Associates, Sunderland, CA, 37–56.
Anderson, DR. 2008. Model-based inference in the life sciences: a primer on evidence. Springer, New York, NY, 184 pp.
Angelini, C. & Silliman, BR. 2014. Secondary foundation species as drivers of trophic and functional diversity: evidence from a tree-epiphyte system. Ecology. 95:185–196.   Arribas, LP., Donnarumma, L., Palomo, MG. & Scrosati, RA. 2014. Intertidal mussels as ecosystem engineers: their associated invertebrate biodiversity under contrasting wave exposures. Mar Biodivers. 44:203–211.  Bäcklund, S., Jönsson, M., Strengbom, J., Frisch, A. & Thor, G. 2016. A pine is a pine and a spruce is a spruce: the effect of tree species and stand age on epiphytic lichen communities. PLoS ONE. 11:e0147004 pp.  Ballantyne, M. & Pickering, CM. 2015. Shrub facilitation is an important driver of alpine plant community diversity and functional composition. Biodivers Conserv. 24:1859–1875.  Beermann, AJ., Ellrich, JA., Molis, M. & Scrosati, RA. 2013. Effects of seaweed canopies and adult barnacles on barnacle recruitment: the interplay of positive and negative influences. J Exp Mar Biol Ecol. 448:162–170.  Bertness, MD., Leonard, GH., Levine, JM., Schmidt, PR. & Ingraham, AO. 1999. Testing the relative contribution of positive and negative interactions in rocky intertidal communities. Ecology. 80:2711–2726.  Boller, ML. & Carrington, E. 2006. In situ measurements of hydrodynamic forces imposed on Chondrus crispus Stackhouse. J Exp Mar Biol Ecol. 337:159–170.  Carrington, E., Grace, SP. & Chopin, T. 2001. Life history phase and the biomechanical properties of the red alga Chondrus crispus (Rhodophyta). J Phycol. 37:699–704.  Chen, LC-M. & McLachlan, J. 1972. The life history of Chondrus crispus in culture. Can J Bot. 50:1055–1060.  Chopin, T., Sharp, G., Belyea, E., Semple, R. & Jones, D. 1999. Open-water aquaculture of the red alga Chondrus crispus in Prince Edward Island, Canada. Hydrobiologia. 398/399:417–425.  Clarke, KR. & Warwick, RM. 2001. Change in marine communities: an approach to statistical analysis and interpretation. PRIMER-E Ltd, Plymouth, 175 pp.
Connell, SD., Foster, MS. & Airoldi, L. 2014. What are algal turfs? Towards a better description of turfs. Mar Ecol Prog Ser. 495:299–307.  Corey, P., Kim, JK., Duston, J., Garbary, DJ. & Prithiviraj, B. 2013. Bioremediation potential of Palmaria palmata and Chondrus crispus (Basin Head): effect of nitrate and ammonium ratio as nitrogen source on nutrient removal. J Appl Phycol. 25:1349–1358.  Dislich, R. & Mantovani, W. 2016. Vascular epiphyte assemblages in a Brazilian Atlantic forest fragment: investigating the effect of host tree features. Plant Ecol. 217:1–12.   Ellrich, JA., Scrosati, RA. & Molis, M. 2015. Predator nonconsumptive effects on prey recruitment weaken with recruit density. Ecology. 96:611–616.   Fernández, Á., Arenas, F., Trilla, A., Rodríguez, S., Rueda, L. & Martínez, B. 2015. Additive effects of emersion stressors on the ecophysiological performance of two intertidal seaweeds. Mar Ecol Prog Ser. 536:135–147.  Garbary, DJ., Tompkins, E., White, K., Corey, P. & Kim, JK. 2011. Temporal and spatial variation in the distribution of life history phases of Chondrus crispus (Gigartinales, Rhodophyta). Algae. 26:61–71.  Gibson, MA. 2003. Seashores of the Maritimes. Nimbus Publishing, Halifax, NS, 346 pp.
Golléty, C., Thiébaut, E. & Davoult, D. 2011. Characteristics of the Ascophyllum nodosum stands and their associated diversity along the coast of Brittany, France. J Mar Biol Assoc U K. 91:569–577.  Guenther, RJ. & Martone, PT. 2014. Physiological performance of intertidal coralline algae during a simulated tidal cycle. J Phycol. 50:310–321.   Guidone, M. & Grace, S. 2010. The ratio of gametophytes to tetrasporophytes of intertidal Chondrus crispus (Gigartinaceae) across a salinity gradient. Rhodora. 112:80–84.  Hammill, E., Atwood, TB. & Srivastava, DS. 2015. Predation threat alters composition and functioning of bromeliad ecosystems. Ecosystems. 18:857–866.  Harley, CDG. 2008. Tidal dynamics, topographic orientation, and temperature-mediated mass mortalities on rocky shores. Mar Ecol Prog Ser. 371:37–46.  Harley, CDG. & O’Riley, JL. 2011. Non-linear density-dependent effects of an intertidal ecosystem engineer. Oecologia. 166:531–541.   Hay, ME. 1981. The functional morphology of turf-forming seaweeds: persistence in stressful marine habitats. Ecology. 62:739–750.  Heaven, CS. & Scrosati, RA. 2008. Benthic community composition across gradients of intertidal elevation, wave exposure, and ice scour in Atlantic Canada. Mar Ecol Prog Ser. 369:13–23.  Hu, Z., Guiry, MD., Critchley, AT. & Duan, D. 2010. Phylogeographic patterns indicate transatlantic migration from Europe to North America in the red seaweed Chondrus crispus (Gigartinales, Rhodophyta). J Phycol. 46:889–900.  Johnson, AS. 2001. Drag, drafting, and mechanical interactions in canopies of the red alga Chondrus crispus. Biol Bull. 201:126–135.   Johnson, SC. & Scheibling, RE. 1987. Structure and dynamics of epifaunal assemblages on intertidal macroalgae Ascophyllum nodosum and Fucus vesiculosus in Nova Scotia, Canada. Mar Ecol Prog Ser. 37:209–227.  Jones, E. & Thornber, CS. 2010. Effects of habitat-modifying invasive macroalgae on epiphytic algal communities. Mar Ecol Prog Ser. 400:87–100.  Kikvidze, Z., Brooker, RW., Butterfield, BJ., Callaway, RM., Cavieres, LA., Cook, BJ., Lortie, CJ., Michalet, R., Pugnaire, FI., Xiao, S., Anthelme, F., Björk, RG., Cranston, BH., Gavilán, RG., Kanka, R., Lingua, E., Maalouf, J-P., Noroozi, J., Parajuli, R., Phoenix, GK., Reid, A., Ridenour, WM., Rixen, C. & Schöb, C. 2015. The effects of foundation species on community assembly: a global study on alpine cushion plant communities. Ecology. 96:2064–2069.   Kim, JH. 1997. The role of herbivory, and direct and indirect interactions, in algal succession. J Exp Mar Biol Ecol. 217:119–135.  Lau, WWY. & Martinez, MM. 2003. Getting a grip on the intertidal: flow microhabitat and substratum type determine the dislodgement of the crab Pachygrapsus crassipes (Randall) on rocky shores and in estuaries. J Exp Mar Biol Ecol. 295:1–21.  Longtin, CM., Scrosati, RA., Whalen, GB. & Garbary, DJ. 2009. Distribution of algal epiphytes across environmental gradients at different scales: intertidal elevation, host canopies, and host fronds. J Phycol. 45:820–827.   Lubchenco, J. 1980. Algal zonation in the New England rocky intertidal community: an experimental analysis. Ecology. 61:333–344.  Lubchenco, J. & Menge, BA. 1978. Community development and persistence in a low rocky intertidal zone. Ecol Monogr. 48:67–94.  Martín, A., Miloslavich, P., Díaz, Y., Ortega, I., Klein, E., Troncoso, J., Aldea, C. & Carbonini, AK. 2016. Intertidal benthic communities associated with the macroalgae Iridaea cordata and Adenocystis utricularis in King George Island, Antarctica. Polar Biol. 39:207–220.   Martinez, AJ. 2003. Marine life of the North Atlantic: Canada to New England. Aqua Quest Publications, Locust Valley, NY, 272 pp.
Matias, MG., Arenas, F., Rubal, M. & Sousa Pinto, I. 2015. Macroalgal composition determines the structure of benthic assemblages colonizing fragmented habitats. PLoS ONE. 10:e0142289 pp.  Matos, J., Costa, S., Rodrigues, A., Pereira, R. & Sousa Pinto, I. 2006. Experimental integrated aquaculture of fish and red seaweeds in northern Portugal. Aquaculture. 252:31–42.  McLachlan, JL. 1991. Chondrus crispus (Irish moss), an ecologically important and commercially valuable species of red seaweed of the North Atlantic Ocean. In : Mauchline J, Nemoto T, editors Marine Biology: Its Accomplishments and Future Prospects. Hokusen-sha, Tokyo, 217–233.
Menge, BA. 1995. Indirect effects in marine rocky intertidal interaction webs: patterns and importance. Ecol Monogr. 65:21–74.  Menge, BA., Foley, MM., Pamplin, J., Murphy, G. & Pennington, C. 2010. Supply-side ecology, barnacle recruitment, and rocky intertidal community dynamics: do settlement surface and limpet disturbance matter? J Exp Mar Biol Ecol. 392:160–175.  Pollock, LW. 1998. A practical guide to the marine animals of northeastern North America. Rutgers University Press, New Brunswick, NJ, 367 pp.
Raffaelli, D. & Hawkins, S. 1999. Intertidal ecology. Springer, Dordrecht, 356 pp.
Sagarin, R. & Pauchard, A. 2010. Observational approaches in ecology open new ground in a changing world. Front Ecol Environ. 8:379–386.  Scrosati, R. & DeWreede, RE. 1998. The impact of frond crowding on frond bleaching in the clonal intertidal alga Mazzaella cornucopiae (Rhodophyta, Gigartinaceae) from British Columbia, Canada. J Phycol. 34:228–232.  Scrosati, R., Garbary, DJ. & McLachlan, J. 1994. Reproductive ecology of Chondrus crispus (Rhodophyta, Gigartinales) from Nova Scotia, Canada. Bot Mar. 37:293–300.  Scrosati, R. & Heaven, C. 2007. Spatial trends in community richness, diversity, and evenness across rocky intertidal environmental stress gradients in eastern Canada. Mar Ecol Prog Ser. 342:1–14.  Scrosati, R. & Mudge, B. 2004. Persistence of gametophyte predominance in Chondrus crispus (Rhodophyta, Gigartinaceae) from Nova Scotia after 12 years. Hydrobiologia. 519:215–218.  Sears, JR. 1998. NEAS keys to the benthic marine algae of the northeastern coast of North America from Long Island Sound to the Strait of Belle Isle. Northeast Algal Society, Dartmouth, MA, 161 pp.
Sept, JD. 2008. A photographic guide to seashore life in the North Atlantic: Canada to Cape Cod. Princeton University Press, Princeton, NJ, 224 pp.
Smith, JR., Vogt, SC., Creedon, F., Lucas, BJ. & Eernisse, DJ. 2014. The non-native turf-forming alga Caulacanthus ustulatus displaces space-occupants but increases diversity. Biol Invasions. 16:2195–2208.  Sokal, RR. & Rohlf, FJ. 2012. Biometry: the principles and practice of statistics in biological research. W. H. Freeman, New York, NY, 937 pp.
Thomas, PA. & Packham, JR. 2007. Ecology of woodlands and forests: description, dynamics and diversity. Cambridge University Press, Cambridge, 528 pp.
Valdivia, N. & Molis, M. 2009. Observational evidence of a negative biodiversity-stability relationship in intertidal epibenthic communities. Aquat Biol. 4:263–271.  Vaselli, S., Bertocci, I., Maggi, E. & Benedetti-Cecchi, L. 2008. Assessing the consequences of sea level rise: effects of changes in the slope of the substratum on sessile assemblages of rocky seashores. Mar Ecol Prog Ser. 368:9–22.  Watt, CA. & Scrosati, RA. 2013a. Bioengineer effects on understory species richness, diversity, and composition change along an environmental stress gradient: experimental and mensurative evidence. Estuar Coast Shelf Sci. 123:10–18.  Watt, CA. & Scrosati, RA. 2013b. Regional consistency of intertidal elevation as a mediator of seaweed canopy effects on benthic species richness, diversity, and composition. Mar Ecol Prog Ser. 491:91–99.  Watt, CA. & Scrosati, RA. 2014. Experimental and mensurative data on the abundance of primary producers and consumers from intertidal habitats in Canada. Ecology. 95:1429 pp. 
|
|















