Effect of substratum types on the growth of assimilators and stolons of Caulerpa okamurae (Bryopsidales, Chlorophyta)
Article information
Abstract
To examine the effects of substratum types on the growth of Caulerpa okamurae, sand surface and sand burial experiments were conducted. Five assimilators (erect fronds) per replicate were cultured for 15 d on the surface of three different treatments: fine sand (200 μm), coarse sand (600 μm), and no sand (control). Also, three stolons and three assimilators were buried by fine grain or coarse grain sands and incubated for 15 d. In both experiments, other culture conditions included 25°C, 30 μmol photons m−2 s−1, and 16 : 8 h L : D (light : dark). In both experiments, stolon + assimilator-, assimilator-, and stolon-weights were measured. Relative growth rates (RGRs) of stolon + assimilator weights ranged from 0.43 to 1.95% d−1 at no sand and fine sand treatment, respectively. RGRs for the weight of stolon + assimilator and new assimilators were significantly greater on the fine- and coarse sand surface than the control. In the burial experiments, RGRs of stolons (4.28% d−1 at coarse sand and 5.57% d−1 at fine sand, respectively) were significantly greater than those of assimilators (1.38% d−1 at fine sand and 1.82% d−1 at coarse sand, respectively). When stolons were buried, RGRs for assimilators were greater at the fine sand than at the coarse sand treatment. On the other hand, RGRs of buried assimilators for total frond weights and for newly produced stolons were significantly greater at the coarse sands than at the fine sands. In conclusion, C. okamurae grew well with all substrates of sands and showed better growth on fine sands than coarse ones. This result suggests that the growth of stolons and assimilators of C. okamurae is stimulated after stable attachment to the sand substrates by rhizophores. In addition, stolons showed higher growth rates than the assimilators in the sand burial states, indicating that stolons are more tolerant to low light than assimilators of C. okamurae.
INTRODUCTION
The genus of Caulerpa Lamouroux includes 104 species distributed in a wide geographical range from temperate to tropical regions (de Gaillande et al. 2017, Zubia et al. 2020, Guiry and Guiry 2022). Some species (e.g., C. taxifolia, C. racemosa, and C. brachypus) are well known invasive species with an effective vegetative propagation mechanism (Boudouresque et al. 1995, Smith and Walters 1999). Asexual reproduction via stolons (a green prostrate axis) and fragmentation increases the spread of population through colonization of new locations, causing serious environmental issues (Khou et al. 2007, Zubia et al. 2020). On the other hand, C. lentillifera and C. racemosa var. tubinata, called ‘sea grapes’ and / or ‘green caviar’, are commercially farmed and have high contents of essential nutrients and various health-promoting compounds (Ratana-Arporn and Chirapart 2006, Matanjun et al. 2009, Guo et al. 2015, de Gaillande et al. 2017, Zubia et al. 2020).
Three Caulerpa species (C. ambigua, C. geminata, and C. okamurae) have been reported in Korea (Shin et al. 2021). The former two species are only observed along the coast of Jeju Island, while C. okamurae shows a broad geographical distribution from Jeju Island to the South Sea to Ulleungdo, East Sea, Korea (Kang 1966, Choi et al. 2008, Lee 2008, Shin et al. 2021). Among these species, C. okamurae is most abundant in terms of production and is considered a biological indicator species for ocean warming (Park et al. 2021). In addition, C. okamurae has received attention as a new resource for human food and cosmetic ingredients (Choi et al. 2000, Hwang et al. 2003, Gao et al. 2019, Shin et al. 2021). The cultivation techniques for this alga have been developed in Korea (Hwang et al. 2003) and in Japan (Morita et al. 2013). Gao et al. (2019) reported that stolons with colorless rhizophores anchoring to substrata have a greater tolerance to high and low temperatures, and low salinity than erect fronds (= assimilators) of C. okamurae (Zubia et al. 2020). A similar phenomenon is also observed in nature. For example, assimilators and stolons grow well from spring to autumn, but the stolons can also survive in winter when assimilators die off (Park et al. 2021).
Caulerpa okamurae may increase biomass and expand geographical distributions using two different vegetative growth patterns; creeping stolon growth adhering several rhizoid clusters (= rhizophores) and upright growth of highly dense assimilators arising from the stolon (Park et al. 2021, Shin et al. 2021). C. okamurae fronds attach to various substrata such as rock, gravel, sand, sponge, and silt in nature. Furthermore, fragmented assimilators and stolons of C. okamurae grow well in the laboratory cultures (Gao et al. 2019, Park et al. 2021, Shin et al. 2021) since fragmented fronds of Caulerpa spp. can grow into new thalli (Smith and Walters 1999). In field populations, stolons and / or assimilators of C. okamurae can be fragmented by environmental factors such as strong wave action and / or herbivory. The C. okamurae fragments can be buried in various sediment types such as silt and / or sand with different grain sizes. Previous studies showed that frond fragments of some Caulerpa species (e.g., C. taxifolia, C. racemosa, and C. lentillifera) can survive, reattach to various substrata, and grow even in the mud (Horstmann 1983, Trono 1990, Ceccherelli and Piazzi 2001, Wright and Davis 2006, Mary et al. 2009). The present study is to examine the effects of substratum types on the growth of C. okamurae and to determine the capacity of regrowth of stolons and assimilators in the sand surface and sand burial states. We hypothesized (1) the presence of sand grain substrata may stimulate the rhizopore development for stable attachment and enhance assimilator and stolon growth of C. okamurae, and (2) creeping stolons, occasionally covered by sediments and exposed to lower light due to dense assimilators in the field, may grow faster than assimilators in the sand substrates.
MATERIALS AND METHODS
Sand grain sizes and frond growth
Caulerpa okamurae fronds were collected from low intertidal rocky shores in Jindo, Jeonnam, Korea (34°39′ N, 126°27′ E) in June 2021. The samples were placed in a cooler and transported to the laboratory. The fronds were washed with filtered seawater to remove muds, detritus, and small animals. Healthy fronds were selected, and the surface was gently brushed with a smooth artist’s brush to reduce epiphytes. More than 70 assimilators of C. okamurae were cut into 3 cm long fragments and pre-cultured at room temperature for 3 d to minimize the wound effect.
The sand grains at the collection site were, in general, a mixture of different sizes of sands from 200 to 600 μm in diameter, covering the hard rocks, where C. okamurae grows. Sand grains collected near the C. okamurae sampling site were washed with distilled water several times and placed in a furnace (J-FM2; JISICO, Seoul, Korea) at 500°C for 1 h to remove organic matter. Two substrate types, fine sand (200 μm) and coarse sand (600 μm) were separated by sieving the sediments. Nine 250 mL beakers were used and each beaker contained 200 mL of PES30 medium (30% of PES) (Provasoli 1968) with three experimental treatments; fine sand, coarse sand, and no sand (control) (n = 3). Approximately 8.5 g of fine or coarse sands were placed in each beaker.
Forty-five assimilators of 70 assimilators were randomly chosen. Five assimilators per beaker were placed on the surface of fine sands, coarse sands, or glass beakers. The assimilators were cultured at 25°C, 30 μmol photons m−2 s−1, and a photoperiod of 16 : 8 h L : D for 15 d. The culture medium of PES30 was changed every 5 d. Stolon and assimilator weights were measured at the initial and final days. At the end of the experiment, stolon + assimilator, stolon, or assimilator weighed with a 3-decimal analytical balance (Model BSA323S-CW; Sartorius, Goettingen, Germany) after blotting excess moisture with absorbent paper. In addition, the number of newly produced assimilators and rhizophores (see Zubia et al. 2020) were counted on day 15. Relative growth rate (RGR% d−1) was calculated based on the wet weight of C. okamurae fronds using the following equation (Choi et al. 2008):
, where W1 and W2 are the initial and final wet weight, respectively, and T is the length of the experiment in days.
Sand burial fronds and growth
To examine the sediment burial effects on the growth of stolons and assimilators, C. okamurae fronds were excised into 4 cm long stolons and 3 cm long assimilators. The excised stolons and assimilators were placed in two glass flasks (1 L), separately and cultured for 3 d for wound recovery. For each replicate, three stolons (ca. 0.50 g in wet weight) or assimilators (ca. 2.05 g) were carefully buried in plant culture dishes (10 cm diameter, 4 cm height) by using two spoons of fine sands or coarse sands (n = 3). Fragmented stolons and assimilators were completely covered by fine- or coarse-sands and incubated for 15 d under the same conditions described above in the sand grain size experiment. The initial and final wet weight for new stolons and assimilators were measured to calculate the RGRs.
Statistical analysis
A one-way ANOVA was applied to evaluate the RGRs of stolons + assimilators, stolons, or assimilators, and the number of newly produced assimilators and rhizoid clusters of C. okamurae. Tukey’s honest significant difference test was applied when significant differences were detected between means (Sokal and Rohlf 1995). Statistical analyses were carried out using STATISTICA ver. 5.0 software (StatSoft Inc., Tulsa, OK, USA).
RESULTS
Sand grain sizes and frond growth
Excised assimilators of Caulerpa okamurae grew well and their weight increased from 2.06 ± 0.01 g (mean ± standard error) at the beginning to 2.20 ± 0.04 g (no sand), 2.47 ± 0.12 g (coarse sand), and 2.78 ± 0.04 g (fine sand) in 15 d. RGRs for total frond weights of C. okamurae were 0.43 ± 0.10 (no sand), 1.25 ± 0.43 (coarse sand), and 1.95 ± 0.12% d−1 (fine sand), respectively (Fig. 1). Stolons + assimilators (total frond) and assimilators only grew better at the fine and coarse sand treatments than control with no sand (Fig. 1). However, the growth rates of newly produced stolons were not significantly different in different substrates.
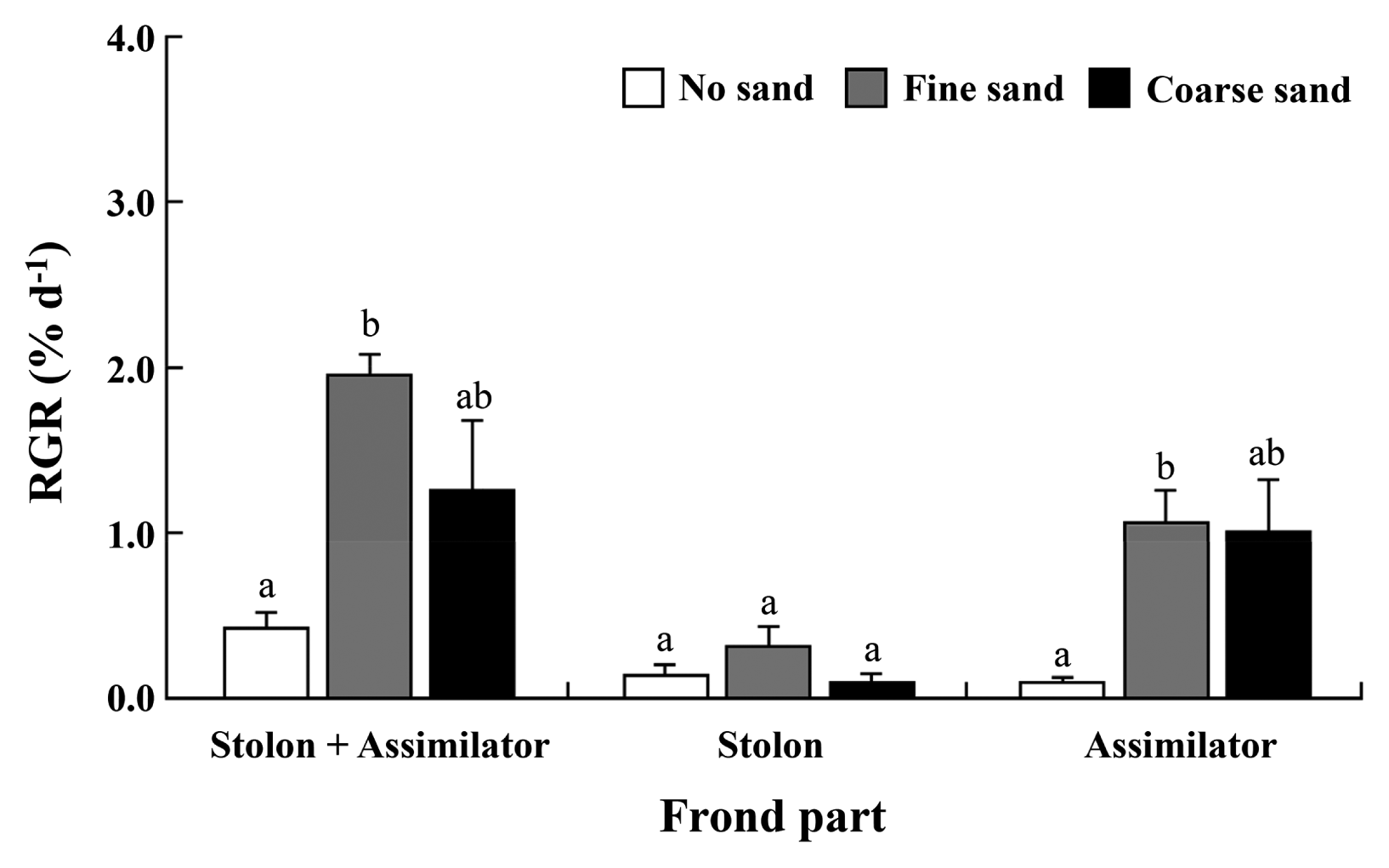
Relative growth rates (RGRs) of assimilators of Caulerpa okamurae grown on different substratum types; no sand, fine- and coarse sands. Assimilators were placed on the surface of each substratum and incubated at 25°C, 30 μmol photons m−2 s−1 and a photoperiod of 16 : 8 h L : D for 15 d. Different letters represent statistical significance (p < 0.05). Data shown are the mean ± standard error (n = 3).
After 15 d, individual assimilator produced rhizophores from 0.83 ± 0.09 (no sand), 1.27 ± 0.07 (coarse sand), to 1.47 ± 0.13 rhizophores (fine sand) and attached to substrata. Production of rhizophores was not significantly different between two different sand grains (Table 1). Also, an experimental assimilator produced new assimilators from 1.07 ± 0.27 (no sand), 2.13 ± 0.37 (fine sand), to 2.87 ± 0.47 individuals (coarse sand) (Table 1). Production of rhizophores and new assimilators was significantly greater at fine and coarse sands than control (Table 1). However, no differences were observed in between fine and coarse sand treatments within 15 d of culture.
Sand burial fronds and their growth
The stolon fragments of C. okamurae were of green color before burial (Fig. 2A). The stolon fronds survived and grew well in the burial conditions with fine and coarse sands. After 15 d, buried stolons in both fine and coarse sands lost their green color but still produced new stolons and assimilators resulting in an increase of frond weight (Fig. 2B & C). RGRs for stolon + assimilator weights were 4.28 ± 0.39% d−1 at coarse sand grains and 5.57 ± 0.44% d−1 at fine sand grains (Fig. 3A). No significant differences were observed between fine sand and coarse sand treatments in RGRs of stolon + assimilator weights and newly produced stolon weights. However, RGRs for newly produced assimilators from original stolon fragments were significantly greater at fine sand than at coarse sand treatment (Fig. 3A).

Stolons of Caulerpa okamurae grown in the fine sand burial. Experimental stolon (4 cm long) with no assimilators at the initial stage (A), buried stolons produced new stolons and assimilators at day 15 (B) and plant culture dish with newly grown stolons and assimilators (C). The samples were cultured in the fine sands at 25°C, 30 μmol photons m−2 s−1 and a photoperiod of 16 : 8 h L : D for 15 d. Scale bars represent: A & B, 3 cm; C, 2 cm.
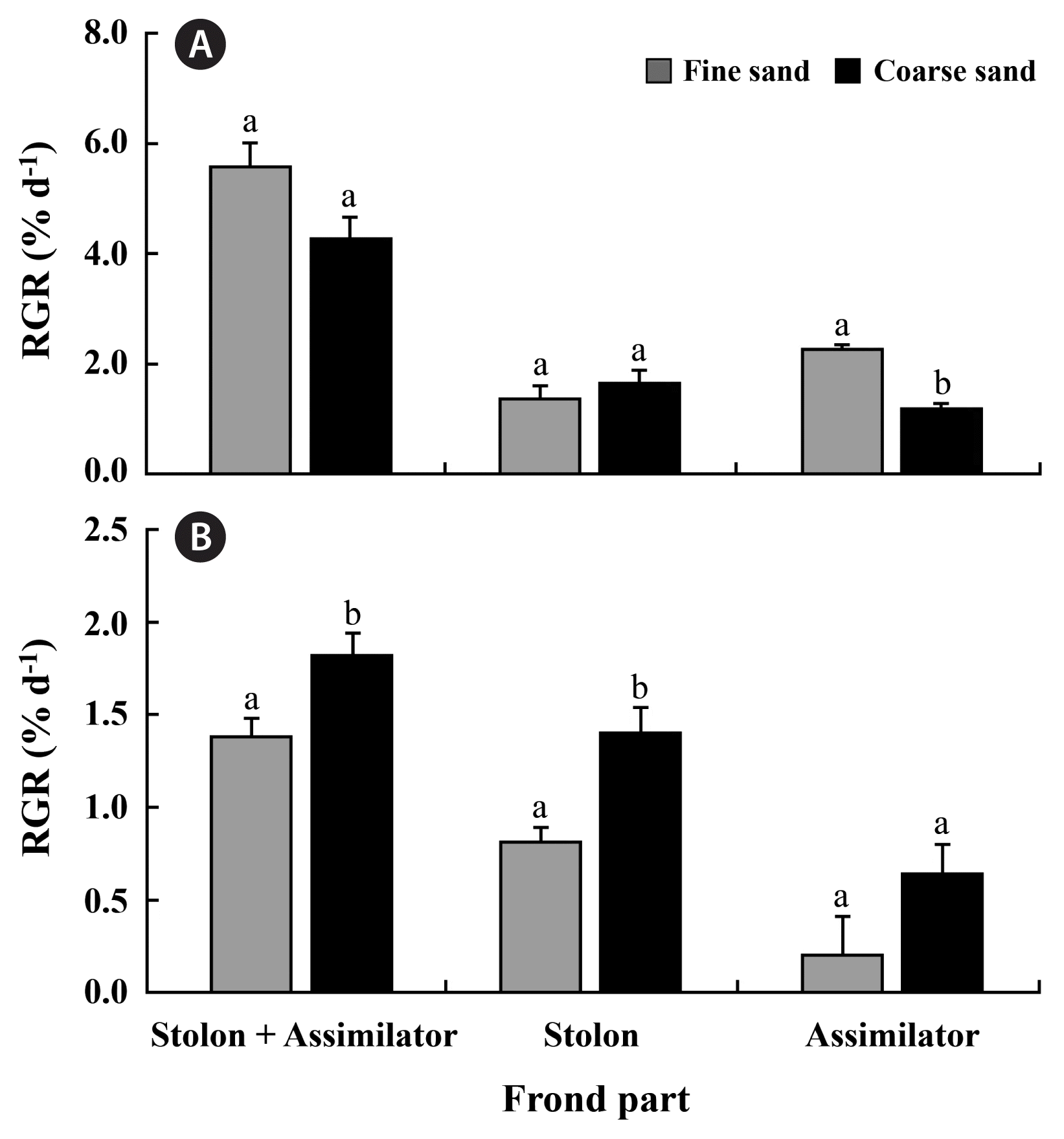
Relative growth rates (RGRs) of stolons + assimlators, stolons, and assimilators weights originated from stolon fragments (A) and assimilator fragments (B) of Caulerpa okamurae buried in fine and coarse sands for 15 d. The samples were incubated at 25°C, 30 μmol photons m−2 s−1 and a photoperiod of 16 : 8 h L : D in burial state with fine and coarse sands. Different letters represent statistical significance (p < 0.05). Data shown are the mean ± standard error (n = 3).
Assimilator fragments buried in fine or coarse sands grew and produced new stolons and assimilators (Fig. 3B). RGRs for stolon + assimilator weights were 1.38 ± 0.21 and 1.82 ± 0.16% d−1 at fine and coarse sand treatments, respectively (Fig. 3B). RGRs for the stolon + assimilator weights and for newly produced stolon weights of C. okamurae were significantly greater in the coarse sand burial treatment than in the fine sand burial ones (Fig. 3B). However, RGRs for newly produced assimilators were not different in different sand treatments after 15 d of culture (Fig. 3B).
DISCUSSION
The stolon and assimilator fragments of C. okamurae fronds survived and grew well on different substrates and in the sand burial conditions, indicating that C. okamurae may expand its geographical distribution by vegetative proliferation. Vegetative growth in terms of stolon extension or fragmentation is very common in Caulerpa (Khou et al. 2007, Alexandre and Santos 2020, Zubia et al. 2020). Vegetative fragmentation is the main mechanism for an invasive species (e.g., Caulerpa taxifolia, C. racemosa, and C. filiformis) to colonize in new coastal areas (Meinesz et al. 1993, Wright and Davis 2006, Khou et al. 2007). Drifting fragments of C. sertularioides thallus can survive for several days before settling on a proper substratum (Fernández-García et al. 2012) and C. prolifera regenerates new vegetative fronds from the basal stolon after dispersal by fragmentation (Collado-Vides 2002). One of invasive species, C. taxifolia has an effective mechanism of spreading basal stolon in short-range and long-distance dispersal of fragments, transported by currents or anchoring systems and fishing nets (Boudouresque et al. 1995).
Caulerpa species grow well in the habitats of soft and hard bottoms (Williams et al. 1985). C. taxifolia can attach to many different types of substrates, such as rocks, sands, mud, seagrass beds including dead rhizomes of seagrasses in the field (Boudouresque et al. 1995). C. prolifera and C. racemosa can colonize a wide range of habitat types, from hard to soft bottoms (Ceccherelli and Piazzi 2001, Collado-Vides 2002). In this study, stolon and assimilator fragments of C. okamurae grew well on the surface or in the burial states of two sizes of sand grains. RGRs of C. okamurae assimilators were significantly greater at fine sand (1.95% d−1) and at coarse sand (1.25% d−1) than control with no sand (0.43% d−1), indicating that sand grains enhance the assimilator growth of this alga. The production of new assimilators was also increased at the fine and coarse sand treatments. These results imply that fine and coarse sand grains may stimulate the rhizophore development for firm assimilator attachment and then the growth in C. okamurae. Optimal conditions for rhizophore development and frond growth of C. prolifera were identical (Friedlander et al. 2006), while large fragments produced longer rhizoids than smaller fragments (Khou et al. 2007). These results suggest the importance of a study looking at the effects of fragment sizes (large, small) and fragment types (stolon, assimilator, stolon + assimilator) on the rhizophore and frond growth to understand the ecology of C. okamurae in the natural environment.
Frond burial in the sediment affects the frond growth in Caulerpa species (Williams et al. 1985). For example, C. sertularioides and C. cupressoides v. lycopodium f. elegans covered by sediment were survived but showed slower growth than uncovered ones (Williams et al. 1985). In the present study, sand burial stolons and assimilators of C. okamurae grew but burial stolon and assimilator parts changed from darker-green to a light-green in color after 15 d. The change of frond color of C. okamurae due to a low photosynthetic rate under a very low light condition in fine and coarse sand grains. The present study suggests that C. okamurae is a shade-adapted species since this alga survived and even grew in the sediments.
Sand burial stolons of C. okamurae grew much faster (5.57% d−1 at fine sand and 4.28% d−1 at coarse sand, respectively) than sand burial assimilators (1.38% d−1 at fine sand and 1.82% d−1 at coarse sand, respectively). It is difficult to make a direct comparison for the growth between stolons and assimilators of C. okamurae because the weights of stolons and assimilators per replicate were different due to different morphological features in this study. However, such differences in the RGRs between stolons and assimilators may be because of the growth patterns of C. okamurae. Thus, the alga begins to grow from stolon extension first and then produces assimilators from the newly grown stolons. Stolon fragments after anchoring in sands start to produce new assimilators and stolons from the experimental stolon, resulting in much faster growth compared to assimilator fragments. Furthermore, this difference may reflect adaptation to the shade environment of stolons which are occasionally covered by sediment or experience low light by dense assimilators of C. okamurae in the field population. The stolons of C. okamurae showed higher growth rates than assimilators under stressful environments, such as high and low temperatures and low salinity in a previous study (Gao et al. 2019) and low light in sand burial state in the present study. Recently, Alexandre and Santos (2020) reported different nutrient acquisition rates between belowground (stolons + rhizophores) and aboveground (assimilators) parts of C. prolifera. Future study for nutrient uptake ability between stolons and assimilators is also required to fully understand the physiological differences between these two frond parts of C. okamurae.
In conclusion, stolon and blade fragments of Caulerpa okamurae thalli survived and grew well on different sand substrates and in the sand burial conditions. Sand burial stolons grew much faster than burial blades of C. okamurae, indicating that stolons exhibit high tolerance to low light. This result suggests that vegetative fragmentation of blades and stolons in C. okamurae is an important growth mechanism to colonize in new areas and to expand its geographical distributions.
ACKNOWLEDGEMENTS
This paper was supported by Wonkwang University, Korea in 2020.
Notes
The authors declare that they have no potential conflicts of interest.
