Protists in hypoxic waters of Jinhae Bay and Masan Bay, Korea, based on metabarcoding analyses: emphasizing surviving dinoflagellates
Article information
Abstract
Hypoxia can indeed impact the survival of protists, which play a crucial role in marine ecosystems. To better understand the protistan community structure and species that can thrive in hypoxic waters, we collected samples from both the surface and bottom waters during the hypoxic period in Jinhae and Masan Bays and the non-hypoxic period in Jinhae Bay. Subsequently, we utilized metabarcoding techniques to identify the protistan species. During hypoxia, with dissolved oxygen concentrations of 0.8 mg L−1 in Jinhae Bay and 1.8 mg L−1 in Masan Bay within the bottom waters, the phylum Dinoflagellata exhibited the highest amplicon sequence variants richness among the identified protist phyla. Following the Dinoflagellata, Ochrophyta and Ciliophora also displayed notable presence. In hypoxic waters of Jinhae and Masan Bays, we identified a total of 36 dinoflagellate species that exhibited various trophic modes. These included one autotrophic species, 14 mixotrophic species, 9 phototrophic species with undetermined trophic modes (either autotrophic or mixotrophic), 2 kleptoplastidic species, and 10 heterotrophic species. Furthermore, the hypoxic bottom water exhibited a greater number of heterotrophic dinoflagellate species compared to the non-hypoxic surface water within the same water column or the non-hypoxic bottom water. Therefore, feeding by mixotrophic and heterotrophic dinoflagellates may be partially responsible for their dominance in terms of the number of species surviving in hypoxic waters. This study not only introduces the initial documentation of 26 dinoflagellate species surviving in hypoxic conditions but also establishes a foundation for a more comprehensive understanding of the ecophysiology of dinoflagellates in hypoxic marine environments.
INTRODUCTION
Hypoxia, typically defined as oxygen levels equal to or less than 2 mg O2 L−1 or 62.5 μM, frequently results from microbial oxygen consumption during the decomposition of organic materials in marine ecosystems (Diaz 2001, Kemp et al. 2009, Sinkko et al. 2013). Over the last 50 years, the global oxygen levels in oceans have annually declined by 2%; thus, deoxygenation has become one of the critical changes in oceans (Schmidtko et al. 2017, Breitburg et al. 2018). Hypoxia can affect the ecophysiology of marine organisms, influencing factors, such as the survival, abundance, development, metabolism, growth, and reproduction (Sampaio et al. 2021). Species tolerating hypoxia may become dominant, whereas species that cannot tolerate hypoxia may disappear or decline (Nilsson and Rosenberg 1994, Riedel et al. 2012). Furthermore, hypoxia can induce alterations in the structure and function of marine ecosystems.
Marine protists encompass a diverse group of single-celled organisms, varying in cell size, and they originate from nearly all branches of the eukaryotic tree of life (Caron et al. 2012, Massana 2015). Marine protists inhabit various marine environments, from surface layers with light penetration, to deep dark zones, and sediments (Ohtsuka et al. 2015, Lim and Jeong 2021). They play diverse roles in marine ecosystems, as primary producers, prey, predators, symbiotic partners, and parasites (Azam et al. 1983, Jeong et al. 2010, Caron et al. 2012, Ok et al. 2018, 2023a). Consequently, marine protists play critical roles in marine biogeochemical cycles and food webs (Mitra et al. 2014, Edgcomb 2016). Moreover, the survival of marine protist species under hypoxic conditions may affect the structure and function of marine ecosystems. However, identifying surviving protist species using microscopy is challenging due to their relatively small size and occasional subtle morphological differences among species in the same genus.
Recently, molecular tools have been developed to efficiently identify very small protists and quantify their abundance (Gran-Stadniczeñko et al. 2019, Min and Kim 2023). Molecular sequencing is one such useful tool for characterizing protistan community structures, which identifies numerous protist taxa in target environments with high sensitivity and taxonomic resolution (Santoferrara et al. 2020, Jang et al. 2022, Kim et al. 2023). Protist community structures and some species surviving in hypoxic waters have been investigated using molecular sequencing in limited regions such as Long Island Sound, USA and Tolo Harbor, Hong Kong (Rocke et al. 2016, Santoferrara et al. 2022). To better understand protist species that can thrive in hypoxic waters and to comprehensively characterize protist community structures in various marine environments, it is essential to conduct DNA sequencing analyses across a broader range of countries and regions.
Dinoflagellates, a major group of protists in marine ecosystems, are ubiquitously present in marine environments (Gran-Stadniczeñko et al. 2019, Jeong et al. 2021, Lee et al. 2021). They play diverse ecological roles, including primary producers, prey, predators, symbionts, and parasites (Hansen 1991, Coats 1999, Stat et al. 2008, Jeong et al. 2010, Ok et al. 2022). Dinoflagellates have three major trophic modes (autotrophy, heterotrophy, and mixotrophy) and serve as various types of prey and predators (Schnepf and Elbrächter 1992, Jeong et al. 2010, Stoecker et al. 2017, Kang et al. 2023, You et al. 2023). They often form red tides or harmful algal blooms (HABs), accounting for 75% of all HAB species, resulting in extensive global damage in marine ecosystems (Smayda 1997, Hallegraeff 2004, Eom et al. 2021, Sakamoto et al. 2021, Ok et al. 2023b). Thus, understanding the ecophysiological characteristics of dinoflagellates is crucial for comprehending the structure and function of marine ecosystems. However, there is limited data available on dinoflagellate species that can thrive in hypoxic conditions. To better understand the structure and function of marine ecosystems in expanding hypoxic conditions, it is crucial to identify dinoflagellate species surviving under hypoxia and elucidate their trophic modes.
Jinhae and Masan Bays, semi-enclosed bays located in the southern part of the Korean Peninsula, are notorious for frequent annual hypoxia from May to September (Lim et al. 2006). Hypoxia in these regions is primarily caused by anthropogenic eutrophication and thermal stratification resulting from the natural sluggish water circulation (Lee et al. 2018). Although the impact of hypoxia on the macrobenthic community in these areas has been elucidated, the effects of hypoxia on the protistan community remain relatively unexplored (Lim et al. 2006); however, diverse protist species have been found in these bays (Jeong et al. 2013). Thus, these bays are ideal regions for exploring the effects of hypoxia on protistan communities.
In this study, surface and bottom seawater samples were collected from Jinhae and Masan Bays during two scenarios: one during observed hypoxia in the bottom water and non-hypoxic conditions in the surface water in August 2023, and the other from Jinhae Bay in August 2022 when hypoxia was not observed in both the surface and bottom waters. Protistan community structures and species surviving under hypoxic conditions, especially dinoflagellate species, were explored using metabarcoding analyses. The present study provides a foundational understanding of protistan community structures in hypoxic environments.
MATERIALS AND METHODS
Sampling and analyses of hydrological properties
One fixed station, SNUJH, was located in Jinhae Bay near Sokcheon Harbor at a water depth of 3 m. Another fixed station, SNUMS, was located in Masan Bay near Masan Harbor at a water depth of 3 m (Fig. 1A)

Map of the study area (A) and vertical profiles of dissolved oxygen (DO, mg L−1), water temperature (°C), salinity, irradiance (μmol photons m−2 s−1), and chlorophyll-a (μg L−1) concentration in Jinhae Bay on Aug 18, 2023 (B), in Masan Bay on Aug 18, 2023 (C), and in Jinhae Bay on Aug 11, 2022 (D). The red-dotted lines indicate the criterion for hypoxia (2 mg L−1). Irradiances in depths were calculated using surface water values and extinction coefficients.
The dissolved oxygen (DO), temperature, salinity, and chlorophyll-a (Chl-a) at depths of 0, 1, 2, and 3 m at each station were measured by deploying a YSI EXO 1 instrument (YSI, Yellow Springs, OH, USA) in Jinhae and Masan Bays on Aug 18, 2023 and additionally in the Jinhae Bay station on Aug 11, 2022. Irradiance in the surface water were measured using a portable illuminance meter (IM-600; Topcon, Tokyo, Japan). The unit of irradiance in the illuminance meter, Lux, was converted to μmol photon m−2 s−1 using the conversion factor provided by Thimijan and Heins (1983). The Secchi depth was measured using a Secchi disk to calculate the extinction coefficient and light intensity at each depth.
Surface waters at each station were collected using a clean bucket, whereas the bottom waters were almost simultaneously collected using a 2.5-L Niskin sampler. For environmental DNA (eDNA), samples of up to 500 mL were filtered using 25-mm glass-fiber (GF)/C membrane filters (Whatman Inc., Clifton, NJ, USA). The filtered membranes were stored in a 2.0 mL tube at −20°C and subsequently transferred to the laboratory.
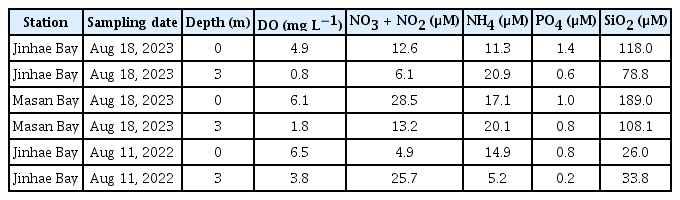
For the nutrient analysis, 20 mL of seawater from each sample was filtered through a GF/F membrane filter (Whatman Inc.). In the laboratory, concentrations of nitrate plus nitrite (NO3 + NO2), ammonium (NH4), phosphate (PO4), and silicate (SiO2) were measured using a nutrient auto-analyzer system (QuAAtro; Seal Analytical GmbH, Norderstedt, Germany).
DNA extraction, sequencing, and sequence analysis
The eDNA collected on the membrane filters were extracted using the AccuPrep genomic DNA extraction kit (Bioneer, Daejeon, Korea). The extracted eDNA was quantified using a Qubit fluorometer using Quant-IT PicoGreen (Invitrogen, Waltham, MA, USA). Subsequently, the extracted eDNA was then stored at −20°C before conducting polymerase chain reaction (PCR).
The sequencing libraries were prepared according to the Illumina metagenomic sequencing library protocols (San Diego, CA, USA) to amplify target genes. The input genomic DNA (5 ng) was amplified using PCR with 5× reaction buffer, 1 mM of deoxyribonucleotide triphosphate (dNTP) mix, 500 nM of universal forward (TAReuk454FWD1; 5′-CCAGCASCYGC GGTAATTCC-3′) and reverse primers (V4 18S Next.Rev; 5′-ACTTTC GTTCTTGATYRATGA-3′) targeting the V4 region of the 18S rRNA gene for protists (Stoeck et al. 2010, Piredda et al. 2017), and Herculase II fusion DNA polymerase (Agilent Technologies, Santa Clara, CA, USA). The first run of PCR included 3 min of incubation at 95°C for heat activation, followed by 25 cycles at 95°C for 30 s, 43°C for 30 s, 72°C for 30 s, and a final extension at 72°C for 5 min. The resulting products were purified using AMPure beads (Agencourt Bioscience, Beverly, MA, USA). Subsequently, 10 μL each of the first PCR products was amplified again with NexteraXT Indexed Primer for the final library construction. The thermocycler settings for the second run of PCR were the same as those for the first run of PCR condition, except for 10 cycles instead of 25 cycles. AMPure beads were used to purify the products. The final purified products were quantified by quantitative PCR (qPCR) according to the qPCR Quantification Protocol Guide (KAPA Library Quantification kits for Illumina Sequencing platforms) and qualified using the TapeStation D1000 ScreenTape (Agilent Technologies, Waldbronn, Germany). The paired-end sequencing was conducted on an Illumina MiSeq platform (Illumina) at Macrogen (Seoul, Korea).
Illumina MiSeq raw data were sorted by sample using index sequences, and paired-end FASTQ files for each sample were generated. The Cutadapt 3.2 tool was used to remove the sequencing adapter sequence and F/R primer sequences of the target gene region (Martin 2011), and then forward (Read 1) and reverse sequences (Read 2) were cut to 230 and 180 bp, respectively. The DADA2 package (version 1.18.0) in R (version 4.0.3) was used to correct errors in amplicon sequencing (Callahan et al. 2016). For paired-end reads, sequences with expected errors of ≥2 were excluded. Then, each sample was denoised after establishing an error model for each batch. After assembling the paired-end sequences into a single sequence, the chimera sequence was removed using DADA2 Consensus method, and then amplicon sequence variants (ASVs) were clustered. Normalization was conducted by subsampling based on the number of sample reads with the minimum number of reads among all samples using the QIIME v. 1.9 program (Caporaso et al. 2010). BLAST+ (version 2.9.0) was performed for taxonomic assignment of each ASV sequence on the reference database (Camacho et al. 2009). If the query coverage of the best hit was less than 85% or the identity was less than 98%, the ASV was not assigned. Moreover, ASVs not assigned to specific species in the PR2 database were further annotated using the National Center for Biotechnology Information (NCBI) database. The ASVs assigned to metazoans were eliminated from the analyses. Raw read sequences were submitted to the NCBI for Biotechnology Information Short Read Archive (accession number: PRJNA1037704).
RESULTS
Hydrographic properties of the study areas
In Jinhae Bay on Aug 18, 2023, with increasing water depth, the DO levels decreased from 4.9 to 0.8 mg L−1 (Fig. 1B). Water temperature also decreased from 28.7 to 26.2°C with increasing water depth; however, salinity increased from 20.8 to 25.9. With increasing water depth, the irradiance decreased from 1,870 to 27 μmol photons m−2 s−1, calculated using an extinction coefficient of 1.42 m−1. Chl-a concentrations also decreased from 58.7 to 3.5 μg L−1. Moreover, the concentrations of NO3 + NO2, PO4, and SiO2 in the surface water were higher than those in the bottom water (Table 1). However, the concentration of NH4 in the surface water was lower than that in the bottom water.
Chemical properties, including dissolved oxygen (DO) and nutrient concentrations, in the surface and bottom waters during each sample collection
In Masan Bay on Aug 18, 2023, with increasing water depth, the DO levels decreased from 6.1 to 1.8 mg L−1 (Fig. 1C). Water temperature also decreased from 28.7 to 25.0°C with increasing water depth; however, salinity increased from 17.7 to 25.8. With increasing water depth, the irradiance decreased from 896 to 30 μmol photons m−2 s−1, calculated using an extinction coefficient of 1.13 m−1. Chl-a concentrations also decreased from 34.3 to 2.8 μg L−1. The concentrations of NO3 + NO2, PO4, and SiO2 in the surface water were higher than those in the bottom water (Table 1). However, the concentration of NH4 in the surface water was lower than that in the bottom water.
In Jinhae Bay on Aug 11, 2022, with increasing water depth, the DO levels decreased from 6.5 to 3.8 mg L−1 (Fig. 1D). With increasing water depth, water temperature slightly decreased from 27.9 to 26.8°C. The salinity of the surface water, 29.5, did not largely differ from that of the bottom water, 30.9. The irradiance at the surface was 476 to 28 μmol photons m−2 s−1, calculated using an extinction coefficient of 1.8 m−1. Chl-a concentrations decreased from 18.3 to 10.6 μg L−1 with increasing water depth. The concentrations of NH4 and PO4 and in the surface water were higher than those in the bottom water (Table 1). However, the concentrations of NO3 + NO2 and SiO2 in the surface water were lower than those in the bottom water (Table 1).
Comparison of protistan community structures in the surface and bottom waters during hypoxic and non-hypoxic periods
In all samples, an average of 55,467 reads (standard error = 4,255) per sample was obtained from the 18S rRNA gene sequencing. In a total of 1,496 ASVs, 27 phyla were annotated (Supplementary Table S1). During the hypoxic period in Jinhae Bay on Aug 18, 2023, the number of ASVs in the hypoxic bottom water, 380, was greater than that in the non-hypoxic surface water, 230 (Fig. 2). Similarly, during the hypoxic period in Masan Bay on Aug 18, 2023, the number of ASVs in the hypoxic bottom water, 412, was greater than that in the non-hypoxic surface water, 354. However, during the non-hypoxic period in Jinhae Bay on Aug 11, 2022, the number of ASVs in the non-hypoxic bottom water, 326, was lower than that in the non-hypoxic surface water, 533. Among the assigned protist phyla in both the surface and bottom waters during both the hypoxic and non-hypoxic periods, the number of ASVs of the phylum Dinoflagellata was the highest, followed by those of the phylum Ochrophyta or Ciliophora (Fig. 2).

The number of amplicon sequence variants, grouped into higher-ranked taxonomic groups in the surface and bottom waters during three sampling events. The red horizontal bars on the top represent the occurrence of bottom-water hypoxia, whereas the blue horizontal bar represents the non-hypoxic event. Numbers on bar graphs indicate the dissolved oxygen concentration (mg L−1) during sample collection.
During the hypoxic period in Jinhae Bay on Aug 18, 2023, the ASVs of all protists detected only in the hypoxic bottom water, 286 (55.4%), were greater than those detected only in non-hypoxic surface water, 136 (26.4%), or those detected in both the surface and bottom waters, 94 (18.2%) (Fig. 3). Similarly, during the hypoxic period in Masan Bay on Aug 18, 2023, the ASVs of all protists detected only in the hypoxic bottom water, 283 (44.4%), were greater than those detected only in the non-hypoxic surface water, 225 (35.3%), or in both the surface and bottom waters, 129 (20.3%). However, during the non-hypoxic period in Jinhae Bay on Aug 11, 2022, the ASVs of all protists detected only in the non-hypoxic surface water, 354 (52.1%), were greater than those detected only in the non-hypoxic bottom water, 147 (21.6%), or in both the surface and bottom waters, 179 (26.3%). The pattern of ASVs of the protists belonging to the phylum Dinoflagellata or phylum Ciliophora was similar to that of all other protists. In contrast, the pattern of ASVs of protists belonging to the phylum Ochrophyta was slightly different from that of all the protists. The ASVs of the phylum Ochrophyta during the hypoxic period in Jinhae Bay on Aug 18, 2023, were highest in both the surface and bottom waters.

Venn diagrams showing the number of amplicon sequence variants (ASVs) of all protists and the dominant phyla detected in the surface water only, bottom water only, and in both the surface and bottom waters. The red horizontal bar on the top represents the occurrence of bottom-water hypoxia, whereas the blue horizontal bar represents the non-hypoxic events.
During the hypoxic period in Jinhae Bay on Aug 18, 2023, in terms of the relative read abundance, both the non-hypoxic surface and hypoxic bottom waters were dominated by the phylum Dinoflagellata (77 and 36%, respectively) (Fig. 4). During the hypoxic period in Masan Bay on Aug 18, 2023, the non-hypoxic surface water was dominated by the phylum Dinoflagellata (49%), while the hypoxic bottom water was dominated by unassigned taxa (59%), followed by Dinoflagellata (15%). During the non-hypoxic period in Jinhae Bay on Aug 11, 2022, both the non-hypoxic surface and bottom waters were dominated by the phylum Ochrophyta (45 and 62%, respectively).

Relative read abundance (%) of protists based on read counts in the surface and bottom waters at three sampling events. The red horizontal bars on the top represent the occurrence of bottom-water hypoxia, whereas the blue horizontal bar represents the non-hypoxic events. Numbers on bar graphs indicate the dissolved oxygen concentration (mg L−1) during sample collection.
Dinoflagellates surviving under hypoxic conditions
The total number of ASVs annotated as the phylum Dinoflagellata in the present study was 301. During the hypoxic period on Aug 18, 2023, the number of ASVs from the phylum Dinoflagellata in the hypoxic bottom waters of both Jinhae and Masan Bays (91 and 86, respectively) was greater than that in the non-hypoxic surface waters (67 and 66, respectively) (Fig. 2). Conversely, in Jinhae Bay, during the non-hypoxic period on Aug 11, 2022, the number of ASVs of Dinoflagellata in the non-hypoxic bottom water (84) was lower than that in the non-hypoxic surface water (119). A total of 73 dinoflagellate species were assigned from the ASV data detected in each sample (Tables 2 & 3). Among these dinoflagellate species, 36 assigned species were detected in the hypoxic waters of Jinhae and Masan Bays, including one autotrophic species, 14 mixotrophic species, nine phototrophic species with undetermined trophic modes (either autotrophic or mixotrophic), two kleptoplastidic species, and 10 heterotrophic species (Table 4).

List of phototrophic dinoflagellates detected in each sample through metabarcoding analysis and their trophic modes (TMs)

List of kleptoplastidic and heterotrophic dinoflagellates detected in each sample through metabarcoding analysis and their trophic modes (TMs)

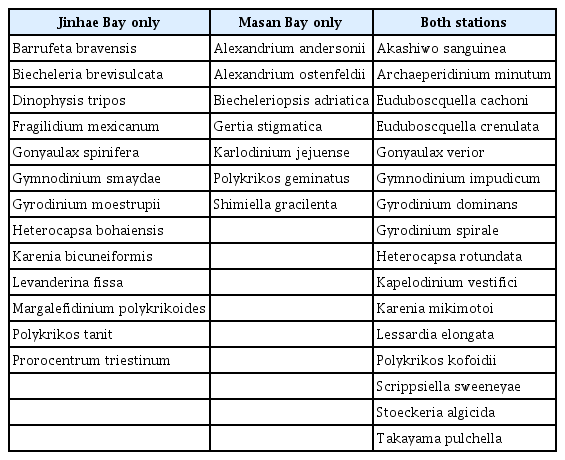
List and the number of the dinoflagellate species surviving in hypoxic waters (<2 mg L−1) in the present study, categorized into two groups: previously known and newly discovered
In Jinhae Bay on Aug 18, 2023, 54 assigned dinoflagellate ASVs (29 assigned dinoflagellate species) that survived in the hypoxic bottom water consisted of one ASV assigned as the autotroph (one autotrophic species), 16 ASVs assigned as mixotrophs (11 mixotrophic species), 20 ASVs assigned as phototrophs (six phototrophic species), one ASV assigned as the kleptoplastidic taxon (one kleptoplastidic species), and 16 ASVs assigned as heterotrophs (10 heterotrophic species) (Fig. 5A & B). Whereas, 51 assigned dinoflagellate ASVs (35 assigned dinoflagellate species) that survived in the non-hypoxic surface water consisted of two ASVs assigned as autotrophs (two autotrophic species), 21 ASVs assigned as mixotrophs (14 mixotrophic species), 19 ASVs assigned as phototrophs (11 phototrophic species), one ASV assigned as the kleptoplastidic taxon (one kleptoplastidic species), and eight ASVs assigned as heterotrophs (seven heterotrophic species).

The number (A & B) and proportion (C & D) of amplicon sequence variants (ASVs) or species identified as autrotrophic, mixotrophic, phototrophic (whose trophic modes have not yet been explored, namely, autotrophic or mixotrophic), kleptoplastidic, and heterotrophic dinoflagellate species in the surface and bottom waters during each sampling date. Venn diagrams (E) showing the number of ASVs or species assigned as Dinoflagellata detected in the hypoxic bottom waters in Jinhae and Masan Bays. Unassigned dinoflagellate taxa were excluded from this analysis. Tables 2 and 3 provide reference information. The red horizontal bars on the top represent the occurrence of bottom-water hypoxia, whereas the blue horizontal bars represent the non-hypoxic events. Numbers on bar graphs indicate the dissolved oxygen concentration (mg L−1) during sample collection.
In Masan Bay, on Aug 18, 2023, 29 assigned dinoflagellate ASVs (23 assigned dinoflagellate species) that survived in the hypoxic bottom water consisted of eight ASVs assigned as mixotrophs (six mixotrophic species), 10 ASVs assigned as phototrophs (seven phototrophic species), one ASV assigned as the kleptoplastidic taxon (one kleptoplastidic species), and 10 ASVs assigned as heterotrophs (nine heterotrophic species) (Fig. 5A & B). Whereas, 46 assigned dinoflagellate ASVs (31 assigned dinoflagellate species) that survived in the non-hypoxic surface water consisted of two ASVs assigned as autotrophs (two autotrophic species), 18 ASVs assigned as mixotrophs (13 mixotrophic species), 22 ASVs assigned as phototrophs (13 phototrophic species), and four ASVs assigned as heterotrophs (three heterotrophic species).
In Jinhae Bay, on Aug 11, 2022, 34 assigned dinoflagellate ASVs (27 assigned dinoflagellate species) that survived in the non-hypoxic bottom water consisted of one ASV assigned as the autotroph (one autotrophic species), 15 ASVs assigned as mixotrophs (nine mixotrophic species), nine ASVs assigned as phototrophs (nine phototrophic species), one ASV assigned as the kleptoplastidic taxon (one kleptoplastidic species), and eight ASVs assigned as heterotrophs (seven heterotrophic species) (Fig. 5A & B). Whereas, 36 assigned dinoflagellate ASVs (35 assigned dinoflagellate species) that survived in the non-hypoxic surface water consisted of eight ASVs assigned as mixotrophs (seven mixotrophic species), 12 ASVs assigned as phototrophs (12 phototrophic species), two ASVs assigned as kleptoplastidic taxon (two kleptoplastidic species), and 14 ASVs assigned as heterotrophs (14 heterotrophic species).
The proportion of heterotrophic dinoflagellate ASVs to total dinoflagellate ASVs in hypoxic bottom waters (30% in Jinhae Bay and 34% in Masan Bay) was higher than that in non-hypoxic surface waters (16% in Jinhae Bay and 9% in Masan Bay) during the hypoxic period on Aug 18, 2023 (Fig. 5C). Conversely, the proportion in the non-hypoxic bottom water (24% in Jinhae Bay) was lower than that in the non-hypoxic surface water (39% in Jinhae Bay) during the non-hypoxic period on Aug 11, 2022 (Fig. 5C). The proportion of heterotrophic dinoflagellate species to total dinoflagellate species in hypoxic bottom waters (34% in Jinhae Bay and 39% in Masan Bay) was higher than that in non-hypoxic surface waters (20% in Jinhae Bay and 10% in Masan Bay) during the hypoxic period on Aug 18, 2023 (Fig. 5D). Conversely, the proportion in the non-hypoxic bottom water (26% in Jinhae Bay) was lower than that in the non-hypoxic surface water (40% in Jinhae Bay) during the non-hypoxic period on Aug 11, 2022 (Fig. 5D).
In the hypoxic bottom waters of Jinhae Bay and Masan Bay, a total of 67 ASVs (36 species) assigned as Dinoflagellata were detected. Among the ASVs (species), 38 ASVs (13 species) were detected in the hypoxic bottom water of Jinhae Bay, whereas 15 ASVs (7 species) were detected in the hypoxic bottom water of Masan Bay. Moreover, 14 ASVs (16 species) were detected in the hypoxic bottom waters of both Jinhae and Masan Bays (Table 5, Fig. 5E).
DISCUSSION
Hydrographic properties of the study areas
Jinhae and Masan Bays are notorious for severe hypoxia from May to October (Lim et al. 2006). These bays face frequent red tides and HABs (Jeong et al. 2013, Ok et al. 2021b, Sakamoto et al. 2021). Red tides often generate substantial amounts of organic material, which can lead to hypoxia (Chen et al. 2021, Dias and Kurian 2022, Ratmaya et al. 2022). When the data obtained in this study were pooled, the DO concentration in the bottom waters was revealed to be inversely related to the Chl-a concentration in the surface waters (Fig. 6). Thus, the high levels of Chl-a in non-hypoxic surface waters may be partially responsible for the formation of hypoxic bottom waters in these study areas.

Bottom dissolved oxygen concentrations (DO) as a function of the surface chlorophyll-a concentration (Chl-a) during the study period.
In these bays, hypoxia was first reported in 2023 on May 23–24 (National Institute of Fisheries Science, Korea; https://www.nifs.go.kr/board/actionBoard0023List.do). In Jinhae Bay, on Aug 16–17, 2023, two days before this study, a hypoxia event with the lowest DO concentration recorded at 0.5 mg L−1 in the bottom water had been reported (National Institute of Fisheries Science, Korea). Therefore, the protists that survived in the hypoxic bottom waters of Jinhae and Masan Bays on Aug 18, 2023, were likely to tolerate hypoxia for at least several days.
Protistan community structures in the hypoxic and non-hypoxic waters
In both Jinhae and Masan Bays, during the hypoxic period, the number of ASVs, (reflective of the species richness) in hypoxic bottom waters was greater than that in the non-hypoxic surface waters. Rocke et al. (2013) reported higher protist species richness in the hypoxic bottom waters than in the non-hypoxic surface waters in the Gulf of Mexico. Conversely, Stauffer et al. (2013) reported lower protist species richness in hypoxic surface water than in non-hypoxic surface water in the northern basin of King Harbor, Southern California Bight. Thus, hypoxia in these regions may not be the only factor affecting ASV or species richness in marine environments. In the present study, during the hypoxic period, the salinities in non-hypoxic surface waters of both Jinhae and Masan Bays were considerably lower than those in the hypoxic bottom waters, indicating an influx of freshwater. Rocke et al. (2013) also reported lower salinities in the non-hypoxic surface water than in the hypoxic bottom water in the Gulf of Mexico, consistent with the results of our study. Conversely, Stauffer et al. (2013) reported little difference in salinities between non-hypoxic and hypoxic surface waters in the northern basin of King Harbor. In general, the number of protist species tends to increase with increasing salinity within the range of 0–35 (Telesh et al. 2013). Thus, during the hypoxic period, the lower salinity of non-hypoxic surface water may partially be responsible for the lower number of ASVs in the non-hypoxic surface water than in the hypoxic bottom water. Moreover, there is a possibility that the ASVs detected in the hypoxic bottom waters in Jinhae and Masan Bays include DNA-containing detritus originating from surface waters, leading to higher numbers of ASVs in the hypoxic bottom waters than in the non-hypoxic surface waters.
Among the phyla that are ASV-assigned in the present study, the phylum Dinoflagellata exhibited the highest relative read abundance in both non-hypoxic surface and hypoxic bottom waters during the hypoxic period. Similarly, Santoferrara et al. (2022) reported that the relative read abundance of the phylum Dinoflagellata in the hypoxic water (<2 mg L−1) of Long Island Sound, USA, was the highest among the protistan phyla. Additionally, Rocke et al. (2016) reported that the relative read abundances of the phylum Dinoflagellata in the hypoxic water of Tolo Harbor, Hong Kong, were the highest and second highest. Therefore, the consistent findings of these three studies indicate that the relative read abundance of the phylum Dinoflagellata in the protistan communities in hypoxic waters was the highest. However, to confirm this hypothesis, the highest relative read abundances of protistan phyla in non-hypoxic and hypoxic waters in more regions should be investigated.
Dinoflagellates surviving in hypoxic waters
The present study found that 36 assigned dinoflagellate species survived in the hypoxic waters using metabarcoding analysis. Among these, 10 dinoflagellate species had been previously reported to be present in hypoxic waters in literature, whereas 26 dinoflagellate species were newly discovered to be present in hypoxic waters in the present study. Thus, the results of the present study largely expanded the roster of dinoflagellate species that can survive in hypoxic waters.
Among the 36 assigned dinoflagellate species that survived in hypoxic waters, 26 were mixotrophic, heterotrophic, and kleptoplastidic, capable of feeding on prey. Therefore, feeding by these dinoflagellates may partially be responsible for their dominance among all the dinoflagellate species surviving in the hypoxia waters of Jinhae and Masan Bays. In the present study, the heterotrophic dinoflagellate Gyrodinium dominans was consistently present in all samples from both surface and bottom waters of Jinhae and Masan Bays in both hypoxic and non-hypoxic periods. Similarly, Santoferrara et al. (2022) also reported a wide distribution of G. dominans in both the surface and bottom waters during hypoxic and non-hypoxic periods in Long Island Sound, USA. In addition, Eom et al. (2024) reported that G. dominans grew in hypoxic waters by feeding on algal prey in laboratory experiments, which supports our field observations. G. dominans is known to feed on the mixotrophic dinoflagellates Akashiwo sanguinea, Heterocapsa rotundata, Karenia mikimotoi, and Margalefidinium polykrikoides (Kang et al. 2020). During the study period, G. dominans co-occurred with these mixotrophic dinoflagellates in hypoxic water. Therefore, G. dominans may survive in hypoxic environments by feeding on prey that survive in these environments.
When the amounts of organic materials increase or remain constant in bottom waters, global warming can exacerbate hypoxia by intensifying the water column stratification, which prevents mixing between the oxygenated surface and hypoxic bottom waters (Rabalais et al. 2009, Bendtsen and Hansen 2013). The results of the present study clearly show that the proportion of the number of heterotrophic dinoflagellate species to the total dinoflagellate species in hypoxic bottom waters increased compared with that in non-hypoxic surface waters during the hypoxic period. Conversely, the proportion in non-hypoxic bottom water decreased compared with that in non-hypoxic surface water during the non-hypoxic period. Thus, more frequent or intensified hypoxia may alter the structure of dinoflagellate communities. Heterotrophic dinoflagellates play diverse roles, such as predators of bacteria, nanoflagellates, diatoms, other dinoflagellates, and eggs and early naupliar stages of copepods, and as prey for diverse metazoans (Strom and Buskey 1993, Jeong 1994, 1999, Sherr and Sherr 2007, Jeong et al. 2008, 2010, 2015). Therefore, hypoxia may eventually affect the structure and function of marine ecosystems.
ACKNOWLEDGEMENTS
We thank Prof. Chung Yeon Hwang at Seoul National University for valuable comments. This research was supported by the National Research Foundation funded by the Ministry of Education (NRF-2022R1A6A3A01086348) award to JHO and the National Research Foundation by the Ministry of Science and ICT (NRF-2021R1A2C1093379; NRF-RS-2023-00291696) and Korea Institute of Marine Science & Technology Promotion (KIMST) by the Ministry of Oceans and Fisheries (20230018) award to HJJ.
Notes
The authors declare that they have no potential conflicts of interest.
SUPPLEMENTARY MATERIALS
Detailed taxonomic information of the protistan communities analyzed by 18S rRNA gene amplicon sequencing during the study period (https://www.e-algae.org).